Amitriptilin
Amitriptilin (Triptomer, Elavil, Triptizol, Laroksil, Saroten, Saroteks, Lentizol, Endep) je triciklični antidepresiv (TCA).[6][7] On je najšire korišćeni TCA i jednako je efikasan protiv depresije kao i novija klasa SSRI lekova.[8] Osim redukovanja simptoma depresije, on olakšava migrene, glavobolje usled napetosti, napade anksioznosti i neke od simptoma šizofrenije. Takođe je poznato da redukuje agresivno i nasilno ponašanje. Neželjena dejstva uključuju: Sedaciju, konfuziju, motornu inkoordinaciju, suva usta, zamućenje vida, posturalna hipotenzija, snižavanje praga konvulzivne aktivnosti, rizik od iznenadne srčane smrti [9]
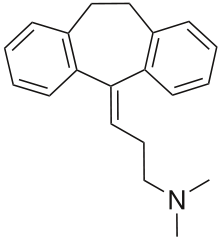
| |||

| |||
| (IUPAC) ime | |||
|---|---|---|---|
| 3-(10,11-dihidro-5H-dibenzo[a,d]ciklohepten-5-iliden)-N,N-dimetilpropan-1-amin | |||
| Klinički podaci | |||
| Robne marke | Triptomer, Elavil | ||
| AHFS/Drugs.com | Monografija | ||
| MedlinePlus | a682388 | ||
| Identifikatori | |||
| CAS broj | 50-48-6 549-18-8 (hidrohlorid) | ||
| ATC kod | N06AA09 | ||
| PubChem[1][2] | 2160 | ||
| DrugBank | DB00321 | ||
| ChemSpider[3] | 2075 | ||
| UNII | 1806D8D52K | ||
| KEGG[4] | D07448 | ||
| ChEBI | CHEBI:2666 | ||
| ChEMBL[5] | CHEMBL629 | ||
| Hemijski podaci | |||
| Formula | C20H23N | ||
| Mol. masa | 277,403 g/mol | ||
| SMILES | eMolekuli & PubHem | ||
| |||
| Farmakokinetički podaci | |||
| Bioraspoloživost | 30–60% usled metabolizma prvog prolaza | ||
| Vezivanje za proteine plazme | > 90% | ||
| Metabolizam | Hepatički CYP2C19, CYP1A2, CYP2D6 | ||
| Poluvreme eliminacije | 10–50 sata, sa prosekom od 15 sata | ||
| Izlučivanje | Renalno | ||
| Farmakoinformacioni podaci | |||
| Trudnoća | D(US) | ||
| Pravni status | ℞ Prescription only | ||
| Način primene | Oralno | ||
Vidi još uredi
Reference uredi
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519.
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Hettne KM, Williams AJ, van Mulligen EM, Kleinjans J, Tkachenko V, Kors JA. (2010). „Automatic vs. manual curation of a multi-source chemical dictionary: the impact on text mining”. J Cheminform 2 (1): 3. DOI:10.1186/1758-2946-2-3. PMID 20331846.
- ↑ Joanne Wixon, Douglas Kell (2000). „Website Review: The Kyoto Encyclopedia of Genes and Genomes — KEGG”. Yeast 17 (1): 48–55. DOI:10.1002/(SICI)1097-0061(200004)17:1<48::AID-YEA2>3.0.CO;2-H.
- ↑ Gaulton A, Bellis LJ, Bento AP, Chambers J, Davies M, Hersey A, Light Y, McGlinchey S, Michalovich D, Al-Lazikani B, Overington JP. (2012). „ChEMBL: a large-scale bioactivity database for drug discovery”. Nucleic Acids Res 40 (Database issue): D1100-7. DOI:10.1093/nar/gkr777. PMID 21948594.
- ↑ Hardman JG, Limbird LE, Gilman AG. (2001). Goodman & Gilman's The Pharmacological Basis of Therapeutics (10 izd.). New York: McGraw-Hill. DOI:10.1036/0071422803. ISBN 0-07-135469-7.
- ↑ Pdr Staff (2009). PDR: Physicians Desk Reference 2010 (Physicians' Desk Reference (Pdr)). Rozelle, N.S.W: Thomson Reuters. ISBN 1-56363-748-0.
- ↑ Barbui C, Hotopf M (February 2001). „Amitriptyline v. the rest: still the leading antidepressant after 40 years of randomised controlled trials”. The British Journal of Psychiatry : the Journal of Mental Science 178 (2): 129–144. DOI:10.1192/bjp.178.2.129. PMID 11157426.
- ↑ James M. Rittter; Rod Flower; Graeme Henderson; Yoon Kong Loke; Humphrey P. Rang. Rang & Dale's Pharmacology (9th izd.). str. 603 - 621.
Spoljašnje veze uredi
- PubChem Substance Summary: Amitriptyline National Center for Biotechnology Information.
- TREPILINE-10 TABLETS; TREPILINE-25 TABLETS South African Electronic Package Inserts. 12 May 1978. Revised February 2004.
- AMITRIP Amitriptyline hydrochloride 10 mg, 25 mg and 50 mg Capsules Medsafe NZ Physician Data Sheet. November 2004.
- Endep Consumer Medicine Information Arhivirano 2008-06-25 na Wayback Machine-u, Australia. December 2005.
- MedlinePlus Drug Information: Amitriptyline. US National Institutes of Health. January 2008.