Klomipramin
(Preusmjereno sa stranice ATC code N06AA04)
Klomipramin (Anafranil) je triciklični antidepresiv (TCA). On je razvijen tokom 1960-tih i od tog vremena je u kliničkoj upotrebi širom sveta.[6][7][8][9]
| |||
| (IUPAC) ime | |||
|---|---|---|---|
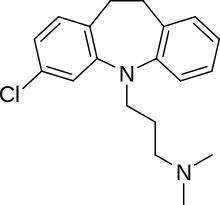
| 3-(3-hloro-10,11-dihidro-5H-dibenzo[b,f]azepin-5-il)-N,N-dimetilpropan-1-amin | |||
| Klinički podaci | |||
| Robne marke | Anafranil | ||
| AHFS/Drugs.com | Monografija | ||
| MedlinePlus | a697002 | ||
| Identifikatori | |||
| CAS broj | 303-49-1 | ||
| ATC kod | N06AA04 | ||
| PubChem[1][2] | 2801 | ||
| DrugBank | DB01242 | ||
| ChemSpider[3] | 2699 | ||
| UNII | NUV44L116D | ||
| KEGG[4] | D07727 | ||
| ChEBI | CHEBI:47780 | ||
| ChEMBL[5] | CHEMBL415 | ||
| Hemijski podaci | |||
| Formula | C19H23ClN2 | ||
| Mol. masa | 314,9 g/mol | ||
| SMILES | eMolekuli & PubHem | ||
| |||
| Farmakokinetički podaci | |||
| Bioraspoloživost | Oralno ~50% | ||
| Metabolizam | Hepatički | ||
| Poluvreme eliminacije | Klomipramin ~35 sata Desmetilklomipramin (glavni aktivni metabolit) ~50 sata | ||
| Izlučivanje | Renalno | ||
| Farmakoinformacioni podaci | |||
| Trudnoća | ? | ||
| Pravni status | ℞ Prescription only | ||
| Način primene | Oralno, IM, IV | ||
Farmakologija uredi
Klomipramin je blokator sledećih transportera:[10]
- Serotoninski transporter (SERT) (Ki = 0.14 nM)
- Norepinefrinski transporter (NET) (Ki = 54 nM)
- Dopaminski transporter (DAT) (Ki = 3,020 nM)
- Glicinski transporter (GlyT/LeuT)
On je antagonist/inverzni agonist sledećih receptora:[10][11][12][13]
- 5-HT2A receptor (Ki = 36 nM)
- 5-HT2C receptor (Ki = 65 nM)
- 5-HT3 receptor (Ki = 85 nM)
- 5-HT6 receptor (Ki = 54 nM)
- 5-HT7 receptor (Ki = 127 nM)
- α1-adrenergički receptor (Ki = 3.2 nM)
- α2-adrenergički receptor (Ki = 525 nM)
- H1 receptor (Ki = 31 nM)
- mACh receptors (Ki = 37 nM)
Aktivni metabolit klomipramina, desmetilklomipramin ispoljava sledeći afinitet:
- Norepinefrinski transporter (NET) (Ki = <1 nM)[14]
Reference uredi
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519.
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Hettne KM, Williams AJ, van Mulligen EM, Kleinjans J, Tkachenko V, Kors JA. (2010). „Automatic vs. manual curation of a multi-source chemical dictionary: the impact on text mining”. J Cheminform 2 (1): 3. DOI:10.1186/1758-2946-2-3. PMID 20331846.
- ↑ Joanne Wixon, Douglas Kell (2000). „Website Review: The Kyoto Encyclopedia of Genes and Genomes — KEGG”. Yeast 17 (1): 48–55. DOI:10.1002/(SICI)1097-0061(200004)17:1<48::AID-YEA2>3.0.CO;2-H.
- ↑ Gaulton A, Bellis LJ, Bento AP, Chambers J, Davies M, Hersey A, Light Y, McGlinchey S, Michalovich D, Al-Lazikani B, Overington JP. (2012). „ChEMBL: a large-scale bioactivity database for drug discovery”. Nucleic Acids Res 40 (Database issue): D1100-7. DOI:10.1093/nar/gkr777. PMID 21948594.
- ↑ Lemke, T.L., Williams, D.A., Roche, V.T., & Zito, S.W., 2012. Foye's Principles of Medicinal Chemistry. 7th ed. Baltimore, MD: Lippincott Williams & Wilkins, p. 605.
- ↑ Healy, D. (2004). Let them eat Prozac: The unhealthy relationship between the pharmaceutical industry and depression. New York: New York University Press, p. 23.
- ↑ Beaumont, G., & Healy, D. (1993). The place of clomipramine in the development of psychopharmacology. Journal of Psychopharmacology, 7, 383–393.
- ↑ Pichot, P. (1996). The discovery of chlorpromazine and the place of psychopharmacology in the history of psychiatry. The Psychopharmacologists (Vol. 1, pp. 1–21). London, England: Chapman & Hall.
- ↑ 10,0 10,1 Millan MJ, Gobert A, Lejeune F, et al. (August 2001). „S33005, a novel ligand at both serotonin and norepinephrine transporters: I. Receptor binding, electrophysiological, and neurochemical profile in comparison with venlafaxine, reboxetine, citalopram, and clomipramine”. The Journal of Pharmacology and Experimental Therapeutics 298 (2): 565–80. PMID 11454918.[mrtav link]
- ↑ Monsma FJ, Shen Y, Ward RP, Hamblin MW, Sibley DR (March 1993). „Cloning and expression of a novel serotonin receptor with high affinity for tricyclic psychotropic drugs”. Molecular Pharmacology 43 (3): 320–7. PMID 7680751.[mrtav link]
- ↑ Ruat M, Traiffort E, Leurs R, et al. (September 1993). „Molecular cloning, characterization, and localization of a high-affinity serotonin receptor (5-HT7) activating cAMP formation”. Proceedings of the National Academy of Sciences of the United States of America 90 (18): 8547–51. DOI:10.1073/pnas.90.18.8547. PMC 47394. PMID 8397408.
- ↑ Toll L, Berzetei-Gurske IP, Polgar WE, et al. (March 1998). „Standard binding and functional assays related to medications development division testing for potential cocaine and opiate narcotic treatment medications”. NIDA Research Monograph 178: 440–66. PMID 9686407.
- ↑ P K Gillman (July 2007). „Tricyclic antidepressant pharmacology and therapeutic drug interactions updated”. Br J Pharmacol. 151 (6): 737–748. DOI:10.1038/sj.bjp.0707253. PMC 2014120. PMID 17471183.