Levonantradol (CP 50,556-1) je sintetički kanabinoidni analog dronabinola (Marinola). Ovaj lek je razvila kompanija Pfizer tokom 1980-tih. On je oko 30x puta potentniji od THC, i manifestuje antiemetske i analgetske efekte putem aktivacije CB1 i CB2 kanabinoidnih receptora.[4] Levonantradol trenutno nije u upotrebi jer su dronabinol i nabilon primenljivi na veći broj oboljenja. On nalazi široku primenu u istraživanju potencijalne terapeutske primene kanabinoida.[5][6][7]
Levonantradol
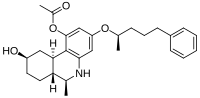
|
| (IUPAC) ime
|
| [(6S,6aR,9R,10aR)- 9-hidroksi- 6-metil- 3-[(2R)-5-fenilpentan- 2-il]oksi- 5,6,6a,7,8,9,10,10a-oktahidrofenantridin- 1-il] acetat
|
| Klinički podaci
|
| Identifikatori
|
| CAS broj
|
71048-87-8
|
| ATC kod
|
nije dodeljen
|
| PubChem[1][2]
|
5361881
|
| ChemSpider[3]
|
4514867
|
| UNII
|
03S640ADSK  Y Y
|
| Hemijski podaci
|
| Formula
|
C27H35NO4
|
| Mol. masa
|
437,571 g/mol
|
| SMILES
|
eMolekuli & PubHem
|
| InChI |
|---|
InChI=1S/C27H35NO4/c1-17(8-7-11-20-9-5-4-6-10-20)31-22-15-25-27(26(16-22)32-19(3)29)24-14-21(30)12-13-23(24)18(2)28-25/h4-6,9-10,15-18,21,23-24,28,30H,7-8,11-14H2,1-3H3/t17-,18+,21-,23+,24-/m1/s1  Y Y
Key: FFVXQGMUHIJQAO-BFKQJKLPSA-N  Y Y |
|
| Farmakoinformacioni podaci
|
| Trudnoća
|
?
|
| Pravni status
|
|
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519. edit
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Hettne KM, Williams AJ, van Mulligen EM, Kleinjans J, Tkachenko V, Kors JA. (2010). „Automatic vs. manual curation of a multi-source chemical dictionary: the impact on text mining”. J Cheminform 2 (1): 3. DOI:10.1186/1758-2946-2-3. PMID 20331846. edit
- ↑ Little PJ, et al. Pharmacology and stereoselectivity of structurally novel cannabinoids in mice. Journal of Pharmacology and Experimental Therapeutics 1988; 247:1046–1051.
- ↑ Tramer MR, et al. Cannabinoids for control of chemotherapy induced nausea and vomiting: quantitative systematic review. British Medical Journal 2001 Jul 7;323(7303):16-21.
- ↑ Campbell FA, et al. Are cannabinoids an effective and safe treatment option in the management of pain? A qualitative systematic review. British Medical Journal. 2001 Jul 7;323(7303):13-6.
- ↑ Ben Amar M. Cannabinoids in medicine: A review of their therapeutic potential. Journal of Ethnopharmacology. 2006 Apr 21;105(1-2):1-25.
Literatura
uredi
- Childers, SR (2006-03-10). „Activation of G-proteins in brain by endogenous and exogenous cannabinoids.”. The AAPS journal 8 (1): E112–7. DOI:10.1208/aapsj080113. PMC 2751429. PMID 16584117.
- Hosking, R.D.; Zajicek, J.P. (2008). „Therapeutic potential of cannabis in pain medicine”. British Journal of Anaesthesia 101 (1): 59–68.
- McCarthy, LE; Borison, HL (1981). „Antiemetic activity of N-methyllevonantradol and nabilone in cisplatin-treated cats.”. Journal of clinical pharmacology 21 (8–9 Suppl): 30S-37S. PMID 6271834.
- Milewich, L; Gant, NF; Schwarz, BE; Chen, GT; MacDonald, PC (1979-03-15). „5 alpha-Reductase activity in human placenta.”. American journal of obstetrics and gynecology 133 (6): 611–7. PMID 34324.
Spoljašnje veze
uredi
