Lekozotan
Lekozotan potencijalni lek za poboljšanje kognitivnih funkcija kod obolelih od Alchajmerove bolesti.[4][5]
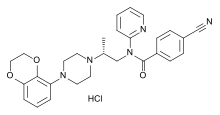
| |||
| (IUPAC) ime | |||
|---|---|---|---|
| 4-Cijano-N-[(2R)-2-[4-(2,3-dihidro-1,4-benzodioksin-5-il)-1-piperazinil]propil]-N-2-piridinilbenzamid hidrohlorid | |||
| Klinički podaci | |||
| Identifikatori | |||
| CAS broj | 434283-16-6 | ||
| ATC kod | nije dodeljen | ||
| PubChem[1][2] | 11156648 | ||
| UNII | 48854OTZ5E | ||
| KEGG[3] | D04683 | ||
| Hemijski podaci | |||
| Formula | C28H30ClN5O3 | ||
| Mol. masa | 520,021 g/mol | ||
| SMILES | eMolekuli & PubHem | ||
| |||
| Farmakoinformacioni podaci | |||
| Trudnoća | ? | ||
| Pravni status | |||
Metod dejstva uredi
Lekozotan je kompetitivni, selektivni antagonist 5-HT1A receptor[6] koji povišava kalijumom stimulisano otpuštanje acetilholina i glutamata.[7]
Reference uredi
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519.
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Joanne Wixon, Douglas Kell (2000). „Website Review: The Kyoto Encyclopedia of Genes and Genomes — KEGG”. Yeast 17 (1): 48–55. DOI:10.1002/(SICI)1097-0061(200004)17:1<48::AID-YEA2>3.0.CO;2-H.
- ↑ H. Spreitzer (2008-08-13). „Neue Wirkstoffe - Lecozotan” (German). Österreichische Apothekerzeitung (17/2007): 805.
- ↑ ClinicalTrials
- ↑ Schlechter, LE; Smith, DL; Rosenzweig-Lipson, S; Sukoff, SJ; Dawson, LA; Marquis, K; Jones, D; Piesla, M i dr.. (2005-06-10). „Lecotozan (SRA-333): A selective serotonin1A receptor antagonist that enhances the stimulated release of glutamate and acetylcholine in the hippocampus and promotes procognitive effects”. Journal of Pharmacology and Experimental Therapeutics 314 (3): 1274. DOI:10.1124/jpet.105.086363. PMID 15951399. Arhivirano iz originala na datum 2007-09-07. Pristupljeno 2014-04-05.
- ↑ Childers, WE Jr, Harrison, BL, Abou-Gharbia, MA, Raje, S, Parks, V, Pangalos, MN, Schechter, LE (2007). „Lecozotan Hydrochloride”. Drugs of the Future 32 (5): 399–407. DOI:10.1358/dof.2007.032.05.1092901.