Umespiron
(Preusmjereno sa stranice BXNRTMZZILHVNJ-UHFFFAOYSA-N)
Umespiron (KC-9172) je lek iz azapironske klase koji poseduje anksiolitičke i antipsihotičke osobine.[5][6][7][8] On je parcijalni agonist 5-HT1A receptora (Ki = 15 nM), D2 receptora (Ki = 23 nM), i antagonist α1-adrenoceptora (Ki = 14 nM). On isto tako ima slab afinitet za sigma receptor (Ki = 558 nM).[5][9][10] Za razliku od drugih anksiolitika i antipsihotika, umespiron proizvodi minimalnu sedaciju, kognitivni deficit, katalepsiju, i ekstrapiramidalne simptome.[4][8][9]
| |||
| (IUPAC) ime | |||
|---|---|---|---|
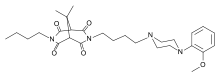
| 3-butil-7-[4-[4-(2-metoksifenil)piperazin-1-il]butil]-9,9-dimetil-3,7-diazabiciklo[3.3.1]nonan-2,4,6,8-tetron | |||
| Klinički podaci | |||
| Identifikatori | |||
| CAS broj | 107736-98-1 | ||
| ATC kod | nije dodeljen | ||
| PubChem[1][2] | 65902 | ||
| ChemSpider[3] | 59311 | ||
| UNII | FG0A3VRL5K | ||
| Hemijski podaci | |||
| Formula | C28H40N4O5 | ||
| Mol. masa | 512,64 g/mol | ||
| SMILES | eMolekuli & PubHem | ||
| |||
| Farmakokinetički podaci | |||
| Poluvreme eliminacije | Nepoznato ali efekti traju znatno duže od drugih azapirona, do 23 nakon jedne doze u ljudskim kliničkim studijama.[4] | ||
| Farmakoinformacioni podaci | |||
| Trudnoća | ? | ||
| Pravni status | nije kontrolisan | ||
| Način primene | oralno | ||
Reference uredi
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519.
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Hettne KM, Williams AJ, van Mulligen EM, Kleinjans J, Tkachenko V, Kors JA. (2010). „Automatic vs. manual curation of a multi-source chemical dictionary: the impact on text mining”. J Cheminform 2 (1): 3. DOI:10.1186/1758-2946-2-3. PMID 20331846.
- ↑ 4,0 4,1 Holland RL, Wesnes K, Dietrich B (1994). „Single dose human pharmacology of umespirone”. European Journal of Clinical Pharmacology 46 (5): 461–8. PMID 7957544.
- ↑ 5,0 5,1 Barnes NM, Costall B, Domeney AM, et al. (September 1991). „The effects of umespirone as a potential anxiolytic and antipsychotic agent”. Pharmacology, Biochemistry, and Behavior 40 (1): 89–96. DOI:10.1016/0091-3057(91)90326-W. PMID 1685786.
- ↑ Ruhland M, Krähling H, Fuchs A, Schön U (November 1988). „KC 9172 (free base of KC 7218)--an antipsychotic/anxiolytic compound. I. Antipsychotic and anxiolytic activity in comparison with chlorpromazine, clozapine, diazepam and buspirone”. Pharmacopsychiatry 21 (6): 396–8. DOI:10.1055/s-2007-1017024. PMID 2907649.
- ↑ Krähling H, Fuchs A, Ruhland M, Schön U, Mol F, Tulp M (November 1988). „KC 9172 (free base of KC 7218)--an antipsychotic/anxiolytic compound. II. Discrimination from typical neuroleptics and benzodiazepine-like minor tranquilizers”. Pharmacopsychiatry 21 (6): 399–401. DOI:10.1055/s-2007-1017025. PMID 2907650.
- ↑ 8,0 8,1 Schmidt WJ, Krähling H, Ruhland M (1991). „Antagonism of AP-5-induced sniffing stereotypy links umespirone to atypical antipsychotics”. Life Sciences 48 (6): 499–505. DOI:10.1016/0024-3205(91)90464-M. PMID 1671523.
- ↑ 9,0 9,1 Ahlenius S, Wijkström A (November 1992). „Mixed agonist-antagonist properties of umespirone at neostriatal dopamine receptors in relation to its behavioral effects in the rat”. European Journal of Pharmacology 222 (1): 69–74. DOI:10.1016/0014-2999(92)90464-F. PMID 1361441.
- ↑ Itzhak Y, Ruhland M, Krähling H (February 1990). „Binding of umespirone to the sigma receptor: evidence for multiple affinity states”. Neuropharmacology 29 (2): 181–4. DOI:10.1016/0028-3908(90)90058-Y. PMID 1970425.