Benzofuranilpropilaminopentan
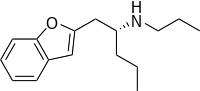
(-)-1-(Benzofuran-2-il)-2-propilaminopentan ((-)-BPAP)[4] je lek sa neobičnim profilom dejstva. On se u izvesnoj meri može grupisanti u familje stimulanata i antidepresiva, mada je njegov mehanizam dejstva veoma različit.[5][6]
| |||
| (IUPAC) ime | |||
|---|---|---|---|
| (2R)-1-(1-Benzofuran-2-il)-N-propilpentan-2-amin | |||
| Klinički podaci | |||
| Identifikatori | |||
| CAS broj | 260550-89-8 | ||
| ATC kod | nije dodeljen | ||
| PubChem[1][2] | 9859674 | ||
| ChemSpider[3] | 7992053 | ||
| Hemijski podaci | |||
| Formula | C16H23NO | ||
| Mol. masa | 245,37 | ||
| SMILES | eMolekuli & PubHem | ||
| |||
| Farmakoinformacioni podaci | |||
| Trudnoća | ? | ||
| Pravni status | |||
Reference uredi
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519.
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Hettne KM, Williams AJ, van Mulligen EM, Kleinjans J, Tkachenko V, Kors JA. (2010). „Automatic vs. manual curation of a multi-source chemical dictionary: the impact on text mining”. J Cheminform 2 (1): 3. DOI:10.1186/1758-2946-2-3. PMID 20331846.
- ↑ US Patent 6214859
- ↑ Shimazu S, Takahata K, Katsuki H, Tsunekawa H, Tanigawa A, Yoneda F, Knoll J, Akaike A. (-)-1-(Benzofuran-2-yl)-2-propylaminopentane enhances locomotor activity in rats due to its ability to induce dopamine release. European Journal of Pharmacology. 2001; 421: 181-189.
- ↑ Shimazu S, Tsunekawa H, Yoneda F, Katsuki H, Akaike A, Janowsky A. Transporter Mediated Actions of R-(-)-1-(Benzofuran-2-yl)-2-propylaminopentane. European Journal of Pharmacology. 2003; 482: 9-16.
Spoljašnje veze uredi
,