Flibanzerin
Flibanserin (BIMT-17, Girosa) je lek koji je razvila kompanija Beringer Ingelhajm kao nehormonski tretman za žene pre menopoze sa poremećajem hipoaktivne seksualne želje.[6][7] Razvoj je prekinut oktobra 2010, nakon što je FDA objavila negativan izveštaj.[8]
| |||
| (IUPAC) ime | |||
|---|---|---|---|
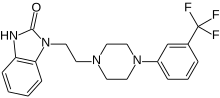
| 1-(24-[3-(trifluorometil)fenil]piperazin-1-il}etil)-1,3-dihidro-2H-benzimidazol-2-on | |||
| Klinički podaci | |||
| Identifikatori | |||
| CAS broj | 167933-07-5 | ||
| ATC kod | nije dodeljen | ||
| PubChem[1][2] | 6918248 | ||
| ChemSpider[3] | 5293454 | ||
| UNII | 37JK4STR6Z | ||
| KEGG[4] | D02577 | ||
| ChEMBL[5] | CHEMBL231068 | ||
| Hemijski podaci | |||
| Formula | C20H21F3N4O | ||
| Mol. masa | 390,40 g/mol | ||
| SMILES | eMolekuli & PubHem | ||
| |||
| Farmakoinformacioni podaci | |||
| Trudnoća | ? | ||
| Pravni status | Nije kontrolisan | ||
| Način primene | Oralno | ||
Reference uredi
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519.
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Hettne KM, Williams AJ, van Mulligen EM, Kleinjans J, Tkachenko V, Kors JA. (2010). „Automatic vs. manual curation of a multi-source chemical dictionary: the impact on text mining”. J Cheminform 2 (1): 3. DOI:10.1186/1758-2946-2-3. PMID 20331846.
- ↑ Joanne Wixon, Douglas Kell (2000). „Website Review: The Kyoto Encyclopedia of Genes and Genomes — KEGG”. Yeast 17 (1): 48–55. DOI:10.1002/(SICI)1097-0061(200004)17:1<48::AID-YEA2>3.0.CO;2-H.
- ↑ Gaulton A, Bellis LJ, Bento AP, Chambers J, Davies M, Hersey A, Light Y, McGlinchey S, Michalovich D, Al-Lazikani B, Overington JP. (2012). „ChEMBL: a large-scale bioactivity database for drug discovery”. Nucleic Acids Res 40 (Database issue): D1100-7. DOI:10.1093/nar/gkr777. PMID 21948594.
- ↑ Borsini F, Evans K, Jason K, Rohde F, Alexander B, Pollentier S. (2002). „Pharmacology of flibanserin”. CNS Drug Rev. 8 (2): 117-142. DOI:10.1111/j.1527-3458.2002.tb00219.x. PMID 12177684.
- ↑ Jolly E, Clayton A, Thorp J, Lewis-D’Agostino D, Wunderlich G, Lesko L (April 2008). „Design of Phase III pivotal trials of flibanserin in female Hypoactive Sexual Desire Disorder (HSDD)”. Sexologies 17 (Suppl 1): S133–4. DOI:10.1016/S1158-1360(08)72886-X.
- ↑ Spiegel online: Pharmakonzern stoppt Lustpille für die Frau, 8 October 2010 (in German)
Vidi još uredi
4
Spoljašnje veze uredi
- Vijagra za žene?
- Marazziti D, Palego L, Giromella A, et al. (June 2002). „Region-dependent effects of flibanserin and buspirone on adenylyl cyclase activity in the human brain”. Int. J. Neuropsychopharmacol. 5 (2): 131-40. DOI:10.1017/S1461145702002869. PMID 12135537.
- Podhorna J, Brown RE (June 2000). „Flibanserin has anxiolytic effects without locomotor side effects in the infant rat ultrasonic vocalization model of anxiety”. Br J Pharmacol 130 (4): 739-746. DOI:10.1038/sj.bjp.0703364. PMC 1572126. PMID 10864879. Arhivirano iz originala na datum 2013-01-06. Pristupljeno 2014-04-05.
- Brambilla A, Baschirotto A, Grippa N, Borsini F (December 1999). „Effect of flibanserin (BIMT 17), fluoxetine, 8-OH-DPAT and buspirone on serotonin synthesis in rat brain”. Eur Neuropsychopharmacol 10 (1): 63-7. DOI:10.1016/S0924-977X(99)00056-5. PMID 10647099.