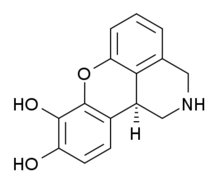
Dinoksilin je sintetičko jedinjenje koje je razvijeno za primenu u naučnim istraživanjima. Dinoksilin deluje kao potentan pun agonist na svih pet podtipova dopaminskog receptora.[3][4][5]
Dinoksilin
|
| (IUPAC) ime
|
| 8,9-dihidroksi-1,2,3,11b-tetrahidrochromeno[4,3,2,-de]isoquinoline
|
| Klinički podaci
|
| Identifikatori
|
| ATC kod
|
nije dodeljen
|
| PubChem[1][2]
|
9819126
|
| Hemijski podaci
|
| Formula
|
C15H13NO3
|
| Mol. masa
|
255,268
|
| SMILES
|
eMolekuli & PubHem
|
| InChI |
|---|
InChI=1S/C15H13NO3/c17-11-5-4-9-10-7-16-6-8-2-1-3-12(13(8)10)19-15(9)14(11)18/h1-5,10,16-18H,6-7H2  Y Y
Key: QOHSTVKJXZTEOL-UHFFFAOYSA-N  Y Y |
|
| Farmakoinformacioni podaci
|
| Trudnoća
|
?
|
| Pravni status
|
|
- ↑ Li Q, Cheng T, Wang Y, Bryant SH (2010). „PubChem as a public resource for drug discovery.”. Drug Discov Today 15 (23-24): 1052-7. DOI:10.1016/j.drudis.2010.10.003. PMID 20970519. edit
- ↑ Evan E. Bolton, Yanli Wang, Paul A. Thiessen, Stephen H. Bryant (2008). „Chapter 12 PubChem: Integrated Platform of Small Molecules and Biological Activities”. Annual Reports in Computational Chemistry 4: 217-241. DOI:10.1016/S1574-1400(08)00012-1.
- ↑ Grubbs RA, Lewis MM, Owens-Vance C, Gay EA, Jassen AK, Mailman RB, Nichols DE (March 2004). „8,9-dihydroxy-1,2,3,11b-tetrahydrochromeno[4,3,2,-de]isoquinoline (dinoxyline), a high affinity and potent agonist at all dopamine receptor isoforms”. Bioorganic & Medicinal Chemistry 12 (6): 1403–12. DOI:10.1016/j.bmc.2004.01.008. PMID 15018913.
- ↑ Ryman-Rasmussen JP, Nichols DE, Mailman RB (October 2005). „Differential activation of adenylate cyclase and receptor internalization by novel dopamine D1 receptor agonists”. Molecular Pharmacology 68 (4): 1039–48. DOI:10.1124/mol.105.012153. PMID 15985612.
- ↑ Zhang J, Xiong B, Zhen X, Zhang A (March 2009). „Dopamine D1 receptor ligands: where are we now and where are we going”. Medicinal Research Reviews 29 (2): 272–94. DOI:10.1002/med.20130. PMID 18642350.
Spoljašnje veze
uredi
